|
Originally he was brought to London, working with the British Tube Alloys nuclear weapons development team. Find the magnetic field which is produced by electrons on protons in the nucleus.Niels Bohr (1885-1962) was a Danish physicist and winner of the 1922 Nobel Prize in Physics.īohr began his work on the Manhattan Project after fleeing to Sweden from Denmark because of German occupation in 1943. Question 2: The electron in Bohr’s Hydrogen atom, revolves around the nucleus with a speed of 2.18 × 10 6 m/s in orbit of radius 5.3 × 10 -11 m. The velocity of an electron in orbit n is given by v n=v 1/n 
Find the velocity of electrons in the second orbit. Question 1: If the velocity of an electron of the first orbit in Bohr’s atomic model of a hydrogen atom is 2.19 × 10 6 m/s. The model couldn’t explain the splitting of spectral lines in the presence of magnetic fields ( Zeeman’s effect ), and electric fields (Stark effect).The model couldn’t explain the presence of fine line structures of different spectral lines of the hydrogen spectrum.The model was applicable to uni electron system only.H v=E f-E i Limitations of Bohr’s Atomic Model In this condition, the energy of photons absorbed is. If E i and E j are the energies of orbits in the initial and final situation, the frequency of the photon emitted will be given by the following equation.Īn electron may jump in a higher energy orbit by absorbing energy from outside. When it happens, a photon is limited whose energy is equal to the difference of energy of two orbits. Energy is emitted from an atom only when an electron jumps from the orbit of higher energy to the orbit having low energy. Third Postulate: The energy of an electron revolving in a stationary orbit remains constant. Therefore the angular momentum “L” of the revolving electron is quantized. Second Postulate: Stationary orbits are those orbits in which the angular momentum of the electron is the integral angular momentum of the electron is an integral multiple of “h/2π”, where “h” is the Planck’s constant. 
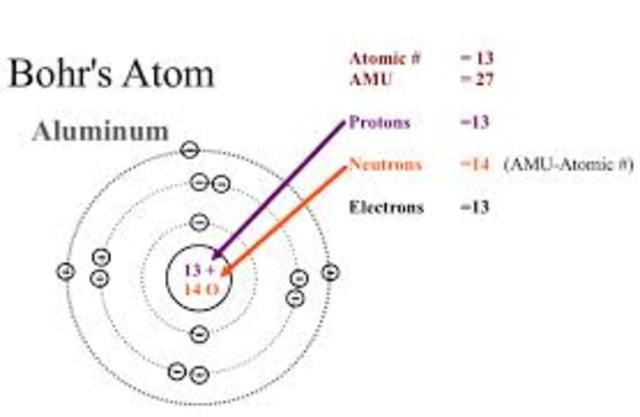
Postulates of Bohr’s atomic modelįirst Postulate: In an atom, electrons revolve around the nucleus in some fixed circular orbits (called stationary orbits), electrons don’t emit energy which was expected from electrodynamics. These planetary models ultimately predicted all atoms to be unstable due to the orbital decay, which was not true and the phenomenon was successfully explained by Bohr’s model. Viewing in terms of electron emission, this would represent a continuum of frequencies being emitted since, as the electron moved closer to the nucleus, it will move faster and will emit a different frequency which was observed experimentally. They used to consider electrons spinning in orbit around a nucleus as a charged particle moving in an electric field, but there was no explanation of the fact that the electron would spiral into the nucleus. The previously proposed planetary models of the atom also had some problems.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
